Research
Exploring the core areas of the Wilson Lab's research program
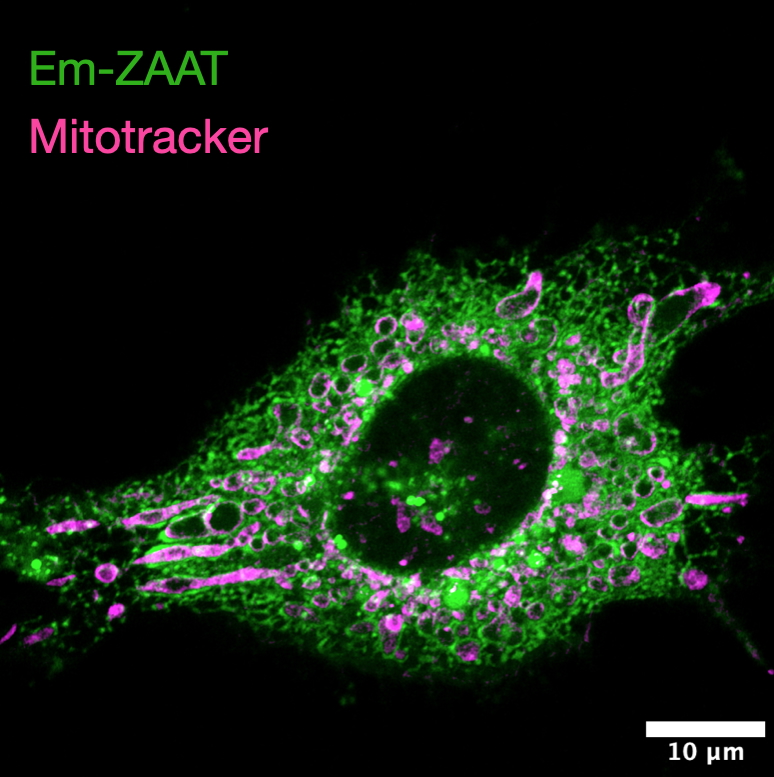
Disease Mechanisms in AATD
We want to define the mechanisms through which misfolded protein aggregates dysregulate lung and liver cells to cause injury and disease. We and others have demonstrated that the differentiated progeny of patient-derived iPSCs model key features of AATD-associated cellular disease. To define a disease signature based on these cells, the Wilson Lab has developed an approach using the CRISPR/Cas9 nuclease to correct the "Z" mutation in patient-derived iPSCs, allowing us to compare isogenic lines derived from a single individual that differ only at the SERPINA1 gene that encodes AAT.
We apply this repository of cells, derived from patients with well-characterized disease outcomes, to generate cell types relevant to lung and liver disease, such as type 2 alveolar epithelial cells, macrophages, and hepatocytes, to observe and define disease mechanisms as they occur in patient cells.
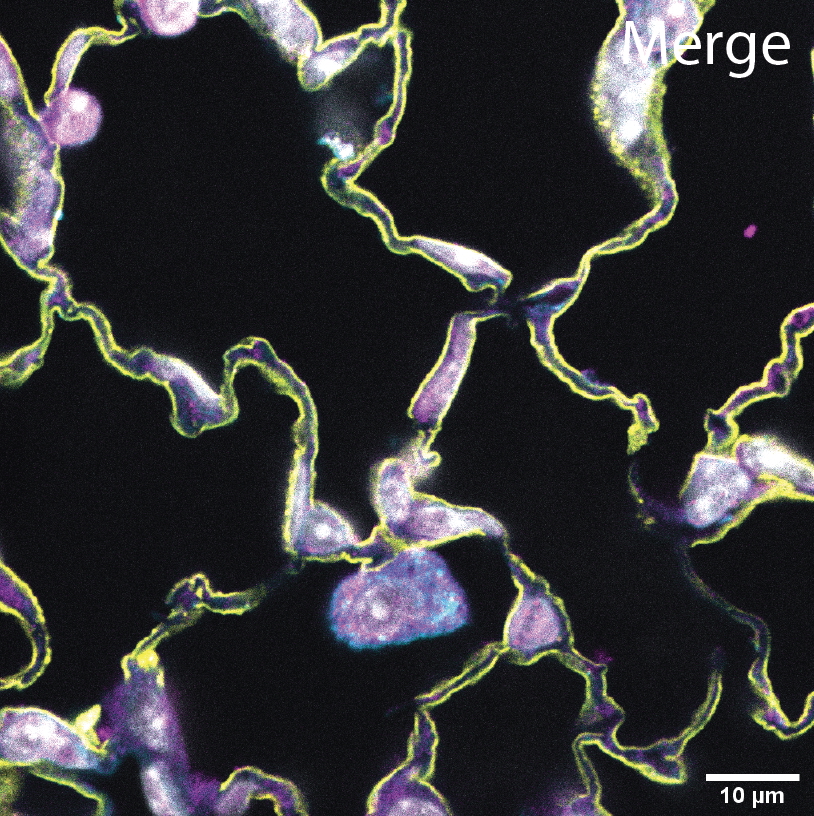
Genetic Drivers of COPD/Emphysema
We work to elucidate the mechanistic contribution of putative COPD susceptibility genes to lung disease pathogenesis. A limited understanding of COPD pathophysiology has hampered the efforts of researchers to develop new potential therapies. COPD develops only in a small percentage of long-term smokers, clusters in families, and exhibits variable prevalence in different racial populations, suggesting a significant contribution of genetic factors to disease susceptibility.
In the past two decades, an increasing number of genes have been identified by GWAS studies as potential contributors to the disease process but their mechanistic contributions to disease remain unknown.
To address this challenge, the Wilson Lab is combining patient iPSCs and CRISPR-based gene manipulation to test the mechanistic contribution of genes associated with COPD risk in GWAS studies to lung development or response to injury.
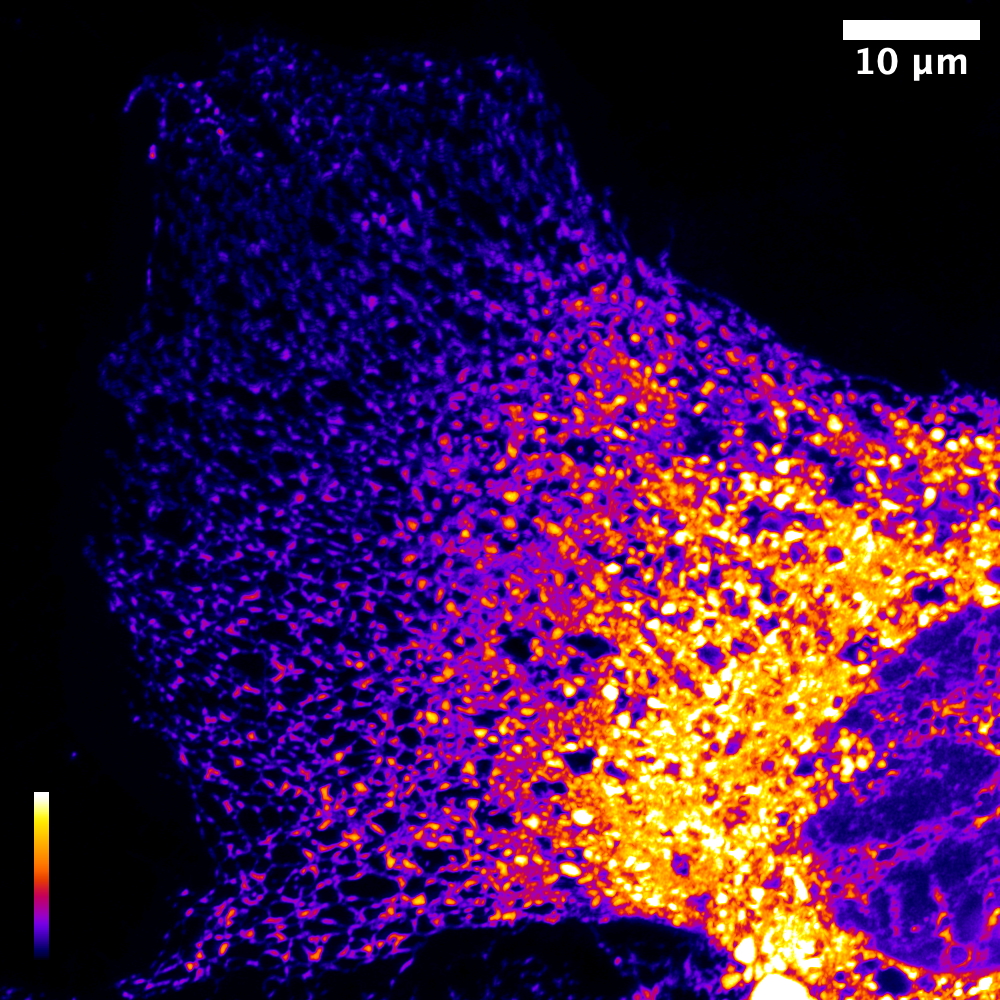
Genetic Factors That Underlie Heterogenous Disease Outcomes
We aim to understand the genetic factors and mechanistic drivers that predispose subsets of AATD patients to develop clinical disease. It is well described that only approximately 15% of individuals homozygous for the single base pair "Z" mutation (homozygotes are referred to as "PiZZ") go on to develop clinically significant liver disease and that genetic cofactors likely contribute to disease susceptibility. Similarly, while PiZZ patients are highly predisposed to developing emphysema, significant heterogeneity exists in the degree of damage observed, with a subset of patients retaining normal lung function throughout their lives.
To identify genes and pathways that contribute to this heterogeneity, we are utilizing CRISPR-based genetic screens in patient-derived cells. We analyze the results of these screens together with sequencing data from patient cohorts to determine potential drivers of specific clinical phenotypes.